Notice that the 'P' abbreviation includes the oxygen atoms and negative charges associated with the phosphate groups. For example, glucose-6-phosphate and isopentenyl diphosphate are often depicted as shown below. 
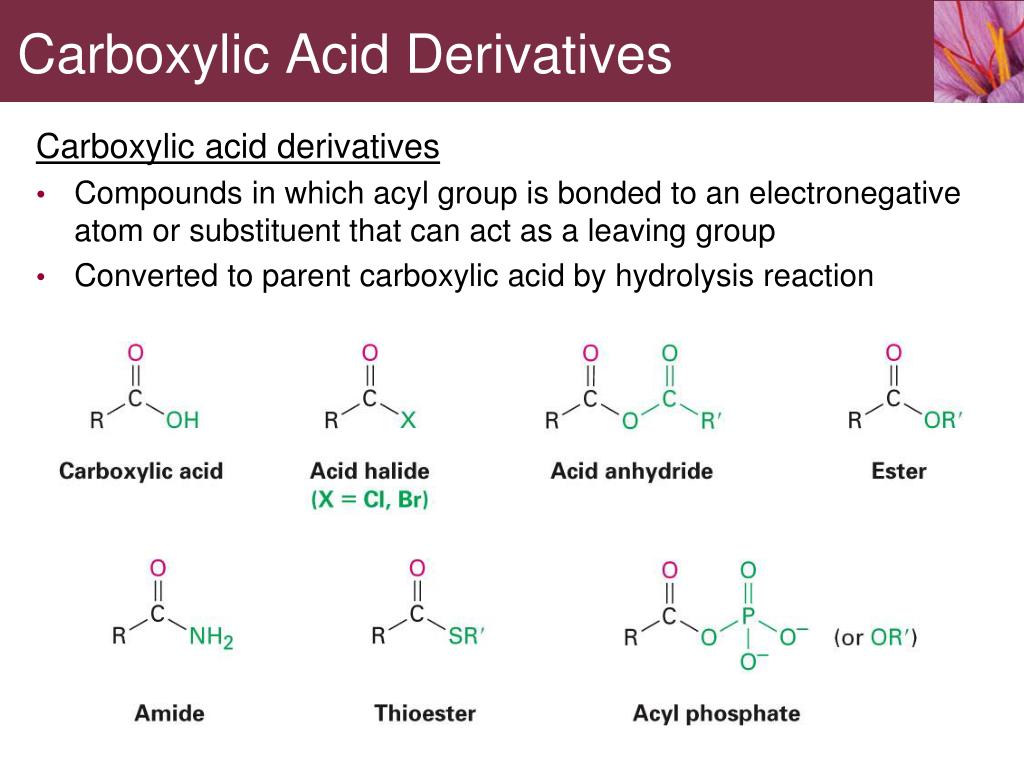
Recall that phosphate groups on organic structures are sometimes abbreviated simply as 'P', a convention that we will use throughout this text. The term 'phosphoryl group' is a general way to refer to all of the phosphate-based groups mentioned in the paragraphs above. The backbone of DNA is composed of phosphate diesters. When a single phosphate is linked to two organic groups, the term 'phosphate diester' is used. An organic diphosphate has two bridging and five non-bridging oxygens. Oxygen atoms in phosphate groups are referred to either 'bridging' and 'non-bridging', depending on their position. Isopententyl diphosphate and adenosine triphosphate (ATP) are good examples: yield a carboxylic acid upon rxn with water. the acyl group, RCO-, bonded to an electronegative atom or substituent, -Y, that can act as a leaving group in substitution reactions (nucleophilic acyl substitution). Then there is a significant drop in reactivity for esters, amides. If an organic molecule is linked to two or three phosphate groups, the resulting species are called organic diphosphates and organic triphosphates. 14.8: Relative Reactivity of Carboxylic Acid Derivatives Halide ions < Acyloxy ions < Alkoxy ions < Amine ions Halide ions > Acyloxy ions > Alkoxy ions >. And Rxns Carboxylic acid derivatives, RCOY or RCOY, can be defined in two ways. Therefore, acid halides are the fastest-reacting derivatives, followed closely by anhydrides.As a general rule, the carbonyl carbon in an acyl group is less electrophilic than that in an aldehyde or ketone. When a phosphate ion is attached to a carbon atom on an organic molecule, the chemical linkage is referred to as a phosphate ester, and the whole species is called an organic monophosphate. The relative reactivity of the carboxylic acid derivatives is an important concept to understand before entering into a detailed examination of nucleophilic acyl substitutions. When two phosphate groups are linked to each other, the linkage is referred to as a 'phosphate anhydride', and the ion is called 'inorganic pyrophosphate' (abbreviation PP i). The fully deprotonated conjugate base of phosphoric acid is called a phosphate ion, or inorganic phosphate (often abbreviated 'P i'). Phosphoryl groups are derivatives of phosphoric acid, a strong acid that is commonly used in the laboratory. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
